Summary
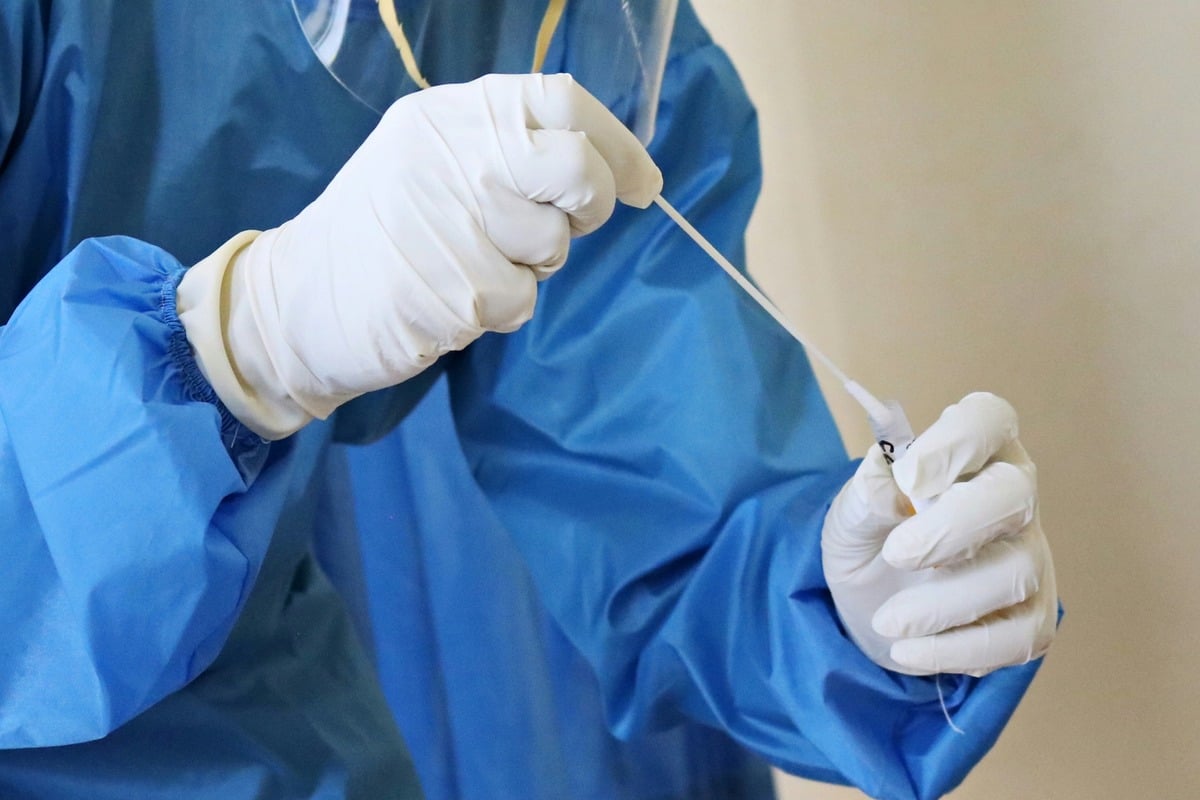
FDA cleared an athome HPV selfcollection kit on April 8; Vanderbilt data show many local cervical cancer cases stem from missed screening.
Source: Hoodline

AI News Q&A (Free Content)
Q1: What are the implications of the FDA's recent clearance of an at-home HPV self-collection kit?
A1: The FDA's clearance of an at-home HPV self-collection kit marks a significant step in increasing accessibility to cervical cancer screening. This development could lead to higher participation rates in screening programs, especially among women who face barriers to accessing traditional healthcare facilities. As cervical cancer is often linked to missed screenings, the availability of a self-collection kit allows for early detection and potentially reduces the incidence of cervical cancer.
Q2: How does the self-sampling method for HPV testing compare to traditional Pap smears in terms of user acceptance?
A2: Research conducted in Kazakhstan has shown that while a significant number of women still prefer gynecologist-collected samples, a notable proportion find self-sampling acceptable. The Evalyn Brush, a type of self-sampling kit, demonstrated higher ease of use and confidence among users compared to the BGI kit. This suggests that with proper education and awareness, self-sampling could become a widely accepted method for HPV testing.
Q3: What challenges do local communities in Nashville face in cervical cancer screening, and how might the at-home HPV test address these?
A3: Vanderbilt data highlight that many cervical cancer cases in Nashville arise from missed screenings. Challenges include limited access to healthcare, socioeconomic barriers, and lack of awareness. The at-home HPV test addresses these issues by providing a convenient, private, and potentially cost-effective option for women who might otherwise forgo screening.
Q4: What are the key factors influencing the acceptance of self-sampling methods for cervical cancer screening according to recent studies?
A4: Studies indicate that ease of use, confidence in the accuracy of self-collection, and the perceived convenience of home-based testing are key factors influencing acceptance. Education level impacts HPV knowledge but not necessarily acceptance of self-sampling. Women who perceive home-based testing as easier than traditional methods are more likely to prefer it for future screenings.
Q5: What role does education play in the acceptance and knowledge of HPV self-sampling methods?
A5: Education plays a crucial role in enhancing HPV knowledge, which in turn can influence the acceptance of self-sampling methods. While higher education levels are associated with increased HPV awareness, studies show that acceptance is more closely tied to individual perceptions of convenience and ease of use rather than education alone.
Q6: How does the introduction of at-home HPV tests align with global health trends in cervical cancer prevention?
A6: The introduction of at-home HPV tests aligns with global health trends focused on increasing accessibility and participation in cervical cancer screening. These tests support efforts to reduce cervical cancer rates, particularly in low- and middle-income countries where traditional screening programs may be less accessible. By empowering women to conduct their own screenings, these tests promote early detection and prevention.
Q7: What are the potential long-term impacts of the FDA's clearance of at-home HPV tests on public health?
A7: The long-term impacts of FDA's clearance of at-home HPV tests could include a significant reduction in cervical cancer incidence and mortality rates due to increased screening coverage. This accessibility can lead to early detection and treatment, ultimately improving public health outcomes and reducing healthcare costs associated with advanced cervical cancer treatment.
References:
- Patient acceptance of HPV self-sampling approach for cervical cancer screening: evidence from a pilot study in Kazakhstan.